RESEARCH
Physical Drivers of Environmental Change
“Our whole earth has become a shore from which we look out across the dark ocean of space, uncertain what we shall find when we sail out among the stars. The stream of time moves forward and mankind moves with it… Mankind is challenged, as it has never been challenged before, to prove its maturity and its mastery; not of nature, but of itself. Therein lies our hope and our destiny.”
Biogeochemistry &
Paleogenomics
Ph.D. Research (2024-Present)

Advisor: Dr. Aradhna Tripati

My current research investigates how marine ecosystems record and respond to environmental change across timescales. Building on established sediment-core and carbonate-proxy approaches in paleoclimate research, I use foraminifera preserved in marine sediments to reconstruct past ocean conditions through clumped isotope geochemistry. This approach enables long-term reconstruction of variability in ocean temperature and carbonate chemistry to track how climate variability has changed the global ocean across timescales from decades to millennia. Alongside this research, I examine how coral biomineralization and symbiotic community structure respond to long-term climate variability and anthropogenic land-use change. My research integrates sub-annual coral geochemistry (Sr/Ca, δ¹⁸O, δ¹¹B, Ba/Ca, Δ¹⁴C) with ancient DNA (coraDNA) metabarcoding of coral symbionts to reconstruct the impacts of ocean warming and acidification on coral reef ecosystems. This research project is one of the first paleoclimate reconstructions to directly link centennial-scale shifts in ocean conditions to changes in Symbiodiniaceae community composition.
M.S. Research (2021-2024)
Quantitative Ecology

Advisor: Dr. Nyssa Silbiger
Intertidal ecosystems are model systems for understanding environmental change because the organisms that inhabit them experience repeated swings in temperature and seawater chemistry that often exceed future climate projections. My previous research examined how ocean warming and acidification interact to influence the physiological energetics of Tegula funebralis, a common intertidal sea snail. Along the Pacific West Coast, Black turban snails are highly abundant and play a key ecological role as a macroalgal grazer; facilitating energy transfer from primary producers to higher trophic levels — a foundational interaction that structures energy flow throughout marine food webs. In dynamic intertidal zones, T. funebralis experiences substantial seasonal and diurnal temperature fluctuations that directly influence metabolic rates (e.g, increased temperatures, increase rates of grazing activity). As oceans continue to warm and acidify due to climate change, understanding how these co-occurring stressors affect the metabolic performance of herbivores is essential for predicting broader ecosystem responses. Therefore, this study asked: How does ocean acidification impact energetic expenditure across a range of ecologically relevant temperatures experienced by T. funebralis?
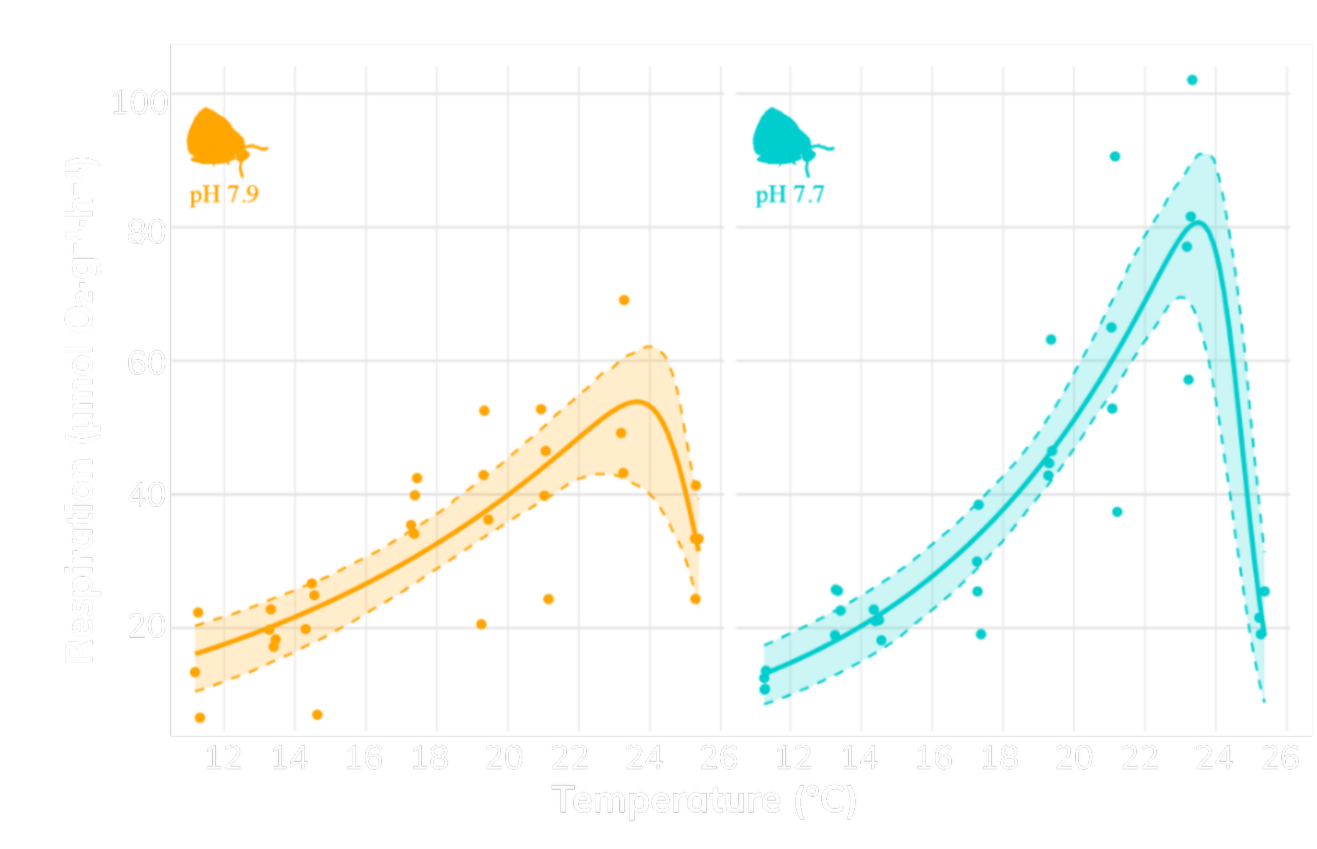
Figure: Comparative thermal performance curves of Tegula funebralis respiration rates (µmol O₂·g⁻¹·h⁻¹) across eight ecologically relevant temperatures (12°C–26°C) and between two seawater pH treatments: ambient conditions (~7.9) and reduced pH (~7.7) simulating ocean acidification. Each point reflects the respiration rate of an individual snail (ambient: n=29; low: n=31), and shaded regions represent bootstrapped 95% confidence intervals around each curve.
To assess how ocean acidification and temperature interactively affect metabolic performance we conducted a mesocosm experiment to compare between two pH levels (ambient ~7.9; reduced ~7.7) across a gradient of eight ecologically relevant temperatures (12°C to 26°C, in 2°C increments). Respiration rates (O₂ consumption), a proxy for energetic expenditure through ATP production were measured after four weeks to generate thermal performance curves. Metabolic responses were then modeled using the Sharpe–Schoolfield equation, which incorporates enzyme kinetics to estimate key physiological parameters, including thermal optima and activation energy, under both ambient and acidified conditions. Results revealed a consistent pattern: sea snails exhibited significantly higher respiration rates near their thermal optimum (~22–24°C) under acidified conditions compared to ambient treatments, indicating that ocean acidification imposes additional metabolic costs even at temperatures typically associated with peak performance. This increase in oxygen demand likely reflects elevated energetic requirements for maintaining acid–base regulation and biomineralization under acidified seawater, where reduced carbonate ion availability (CO₃²⁻) constrains the chemical conditions necessary for shell formation. Together, these findings demonstrate that ocean acidification reshapes thermal performance by elevating metabolic demand, thereby constraining energy budgets and increasing the costs of maintaining essential physiological processes in a rapidly warming ocean.

EARLY RESEARCH EXPERIENCE
Bay Lab, UC Davis (2019-2021)
Population Genomics & Evolution

Conducted a collaborative meta-analysis of reef-building coral microsatellites to evaluate the physical drivers of genetic diversity across the globe.
Advisor: Dr. Rachael Bay
Gold Lab & UC Davis BML (2019)
Geobiology & Environmental Toxicology
Investigated polyp regeneration in moon jellyfish exploring cellular mechanisms of immortality & independent research on microplastic ingestion in urchin larvae
Advisors: Drs. David Gold & Gary Cherr
Study Abroad, Guatemala (2017)
Wetland Ecology & Community Science

Analyzed the role of native versus invasive plant assemblage compositions on water quality parameters in wetland ecosystems in Lake Atitlán.
Advisor: Dr. Eliška Rejmánková

